The valence electrons are held closer towards the nucleus of the atom. This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. The effect of increasing proton number is greater than that of the increasing electron number therefore, there is a greater nuclear attraction. Periodic Trends in the Electronegativities of Elements. However, at the same time, protons are being added to the nucleus, making it more positively charged. 
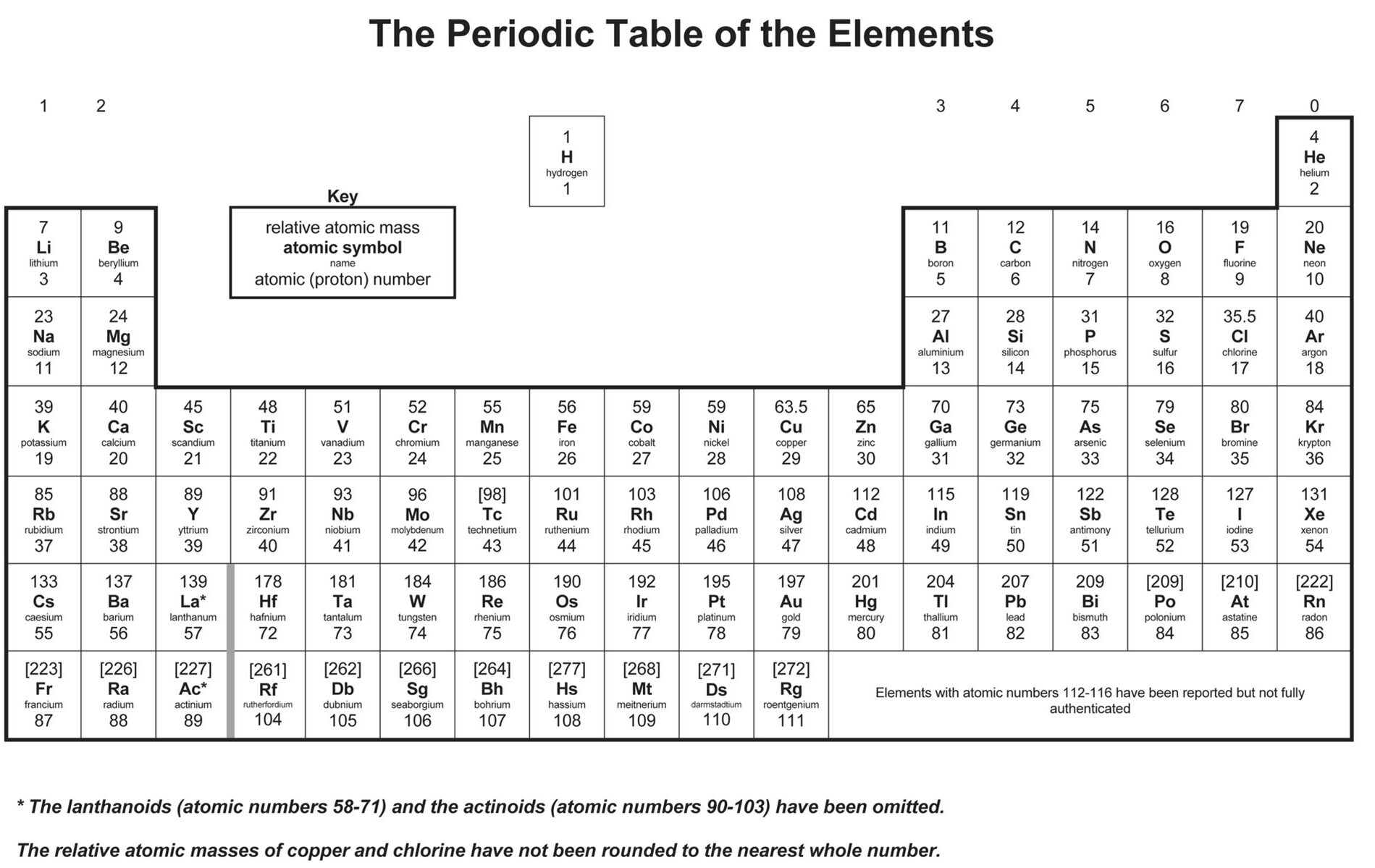
This is because, within a period or family of elements, all electrons are added to the same shell. Atomic radius patterns are observed throughout the periodic table.Ītomic size gradually decreases from left to right across a period of elements. The covalent radii of these molecules are often referred to as atomic radii. KS3 Chemistry Periodic table learning resources for adults, children, parents and teachers. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Metals and non-metals can be heated in oxygen to make compounds called oxides. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. Varying the number of electrons in an atom of an element produces ions. For example, a hydrogen atom has 1 proton, while a carbon atom has 6 protons. Each element has a unique number of protons in its atom. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). A chemical element is a substance that cannot be further broken down by any chemical reaction. This is caused by the increase in atomic radius. Periodicity is caused by regular and predictable variations in element atomic structure. Electron affinity decreases from top to bottom within a group. In the context of chemistry and the periodic table, periodicity refers to trends or recurring variations in element properties with increasing atomic number.ThoughtCo explains how to identify nonmetals on the periodic table, what are their common uses and examples, and how they differ from metalloids and gases. The period number increases by one for every additional row, up to a maximum of 7. Nonmetals are elements that have different properties from metals, such as low conductivity, high electronegativity, and poor heat transfer. Each period is given a numerical value, beginning with '1,' which is assigned to the top row. Valence electrons are outer shell electrons for main group elements. A period is a horizontal (left-to-right) row on the periodic table. mixture When two or more compounds or elements are present without. In chemistry and physics, a valence electron is an electron associated with an atom that can form a chemical bond and participate in a chemical reactions. This is caused by the decrease in atomic radius. compound A pure substance made from two or more elements which are chemically bonded in a fixed ratio. Electron affinity increases from left to right within a period.This causes the electron to move closer to the nucleus, thus increasing the electron affinity from left to right across a period. 
Moving from left to right across a period, atoms become smaller as the forces of attraction become stronger. With a larger distance between the negatively-charged electron and the positively-charged nucleus, the force of attraction is relatively weaker. This means that an added electron is further away from the atom's nucleus compared with its position in the smaller atom. \): Antimony.\( \newcommand\): Periodic Table showing Electron Affinity TrendĮlectron affinity generally decreases down a group of elements because each atom is larger than the atom above it (this is the atomic radius trend, discussed below).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
